LIFEPAK: A Lifesaving Defibrillator Series That Changed Emergency Medicine
Introduction
LIFEPAK is a well-known name in emergency medicine. It is a series of advanced vital signs monitors and external defibrillators created by Physio-Control, a medical technology company based in Redmond, Washington. Over the decades, LIFEPAK devices have saved countless lives in hospitals, ambulances, and even public spaces.
These devices are trusted worldwide because they combine portability, reliability, and innovative technology. From the first LIFEPAK defibrillator in the late 1960s to today’s touchscreen-based LIFEPAK 35, the series continues to set standards in cardiac emergency care.
What is Lifepak?
Lifepak (stylized LIFEPAK) is a series of advanced vital signs monitors and external cardiac defibrillators developed by medical technology company Physio-Control. These devices are designed to monitor heart activity, detect life-threatening arrhythmias, and deliver life-saving shocks when needed.
Lifepak defibrillators are used by healthcare professionals, such as paramedics and emergency medical technicians, as well as by trained members of the public in public access settings.
Over the decades, Lifepak has evolved from the original Lifepak 33 into sophisticated models like the Lifepak 35, incorporating features such as CPR guidance, 12-lead ECG monitoring, and touchscreen controls to support faster and more effective emergency response.
The Beginning of LIFEPAK

The story of LIFEPAK began in 1968, when Physio-Control demonstrated its first LIFEPAK defibrillator, the LIFEPAK 33.
This model weighed 34 pounds, making it the lightest defibrillator of its time. Development took just 90 days, which earned it the nickname “the 90-day wonder.”
By 1969, it became commercially available and quickly gained attention in the medical field. The number “33” in its name came from the company’s original goal of designing a unit that weighed only 33 pounds.
Growth and Innovation Through the Years
Physio-Control continued to innovate, releasing multiple LIFEPAK models over the years.
- 1971: The LIFEPAK 911 introduced 12-lead ECG monitoring.
- 1972: The LIFEPAK 2 allowed ECG transmissions over telephone lines, connecting ambulances with hospitals.
- 1973–1974: Models like the LIFEPAK 3 and LIFEPAK 4 added features such as “freeze” displays and integrated ECG recorders.
- 1976: The LIFEPAK 5 arrived, weighing just 5 pounds and 19 ounces. A modified version even went on the Mount Everest expeditions in 1981 and 1982.
By the late 1980s, models such as the LIFEPAK 9 and 10 introduced advanced documentation, including the ability to record when shocks were given and when medications were administered.
Expanding Access to the Public
In the 1990s, Physio-Control shifted focus toward accessibility.
- 1994: The LIFEPAK 11 improved prehospital ECG transmission.
- 1997: The LIFEPAK 500 was released as a public access defibrillator, making AEDs available for everyday use outside hospitals.
- 1998: The LIFEPAK 12 became popular for its reliable monitoring and diagnostic abilities. Some ambulances still use it today.
Modern Advancements
The 2000s brought even more progress:
- 2002: The LIFEPAK 20 was released for hospitals, while the CR Plus targeted public spaces.
- 2006: The LIFEPAK 1000 replaced the 500 as a professional AED.
- 2008: The LIFEPAK 15 introduced a color LCD, Bluetooth connectivity, and advanced monitoring options.
Finally, in 2024, the LIFEPAK 35 was launched. This model marked a complete redesign, using a touchscreen instead of most hard buttons. It includes CPRINSIGHT technology, pediatric AED protocols, and live ECG monitoring with up to 15 leads.
Who Produces Lifepak?
Lifepak devices are produced by Physio-Control, a renowned medical technology company headquartered in Redmond, Washington. Physio-Control specializes in life-saving medical equipment, focusing on defibrillators, vital signs monitors, and other emergency care solutions.
The company has decades of experience in designing reliable, high-performance devices for both professional medical personnel and trained public users. Lifepak has become one of their flagship product lines, trusted worldwide in prehospital, hospital, and public access environments.
Is Lifepak Produced According to Standards?

Yes, Lifepak defibrillators and monitors are manufactured according to strict medical device standards and regulations. Physio-Control ensures that each Lifepak model complies with international safety, quality, and performance requirements. The devices undergo rigorous testing, including electrical safety, shock delivery accuracy, and functionality under extreme conditions.
Advanced models like the Lifepak 15 and 35 also integrate features such as CPR guidance, ECG monitoring, and diagnostic tools, all developed in line with recognized medical protocols. This adherence to standards ensures that Lifepak devices are both safe and effective for emergency responders and public users alike.
Key Features of LIFEPAK Devices
LIFEPAK defibrillators are not limited to delivering shocks. Many models also include:
- Heart rate monitoring
- 12-lead ECG acquisition and interpretation
- CPR guidance with metronome and verbal prompts
- Synchronized cardioversion
- External pacing
- Oxygen saturation and end-tidal CO₂ monitoring
- Non-invasive and invasive blood pressure monitoring
Advanced models such as the LIFEPAK 15, 20, and 35 are mainly used by paramedics and hospital professionals. Public-friendly AEDs like the LIFEPAK CR2 are designed for trained community members.
Lifepak Defibrillator Models
Over the decades, Lifepak has introduced a wide range of defibrillators designed for both medical professionals and the general public. Early models such as the Lifepak 33, 2, and 5 focused on portability and basic monitoring. As technology advanced, the Lifepak 9, 10, and 12 added features like improved ECG recording and better diagnostic tools. Public access defibrillators such as the Lifepak 500, 1000, and CR Plus made it possible for trained community members to act in emergencies.
Today, professional models like the Lifepak 15, 20, and 35 are used in hospitals and ambulances, offering advanced monitoring, wireless data transmission, and CPR guidance. This wide product range ensures that Lifepak has a solution for almost every setting—from homes and schools to emergency response units.
Who Can Use a Lifepak?
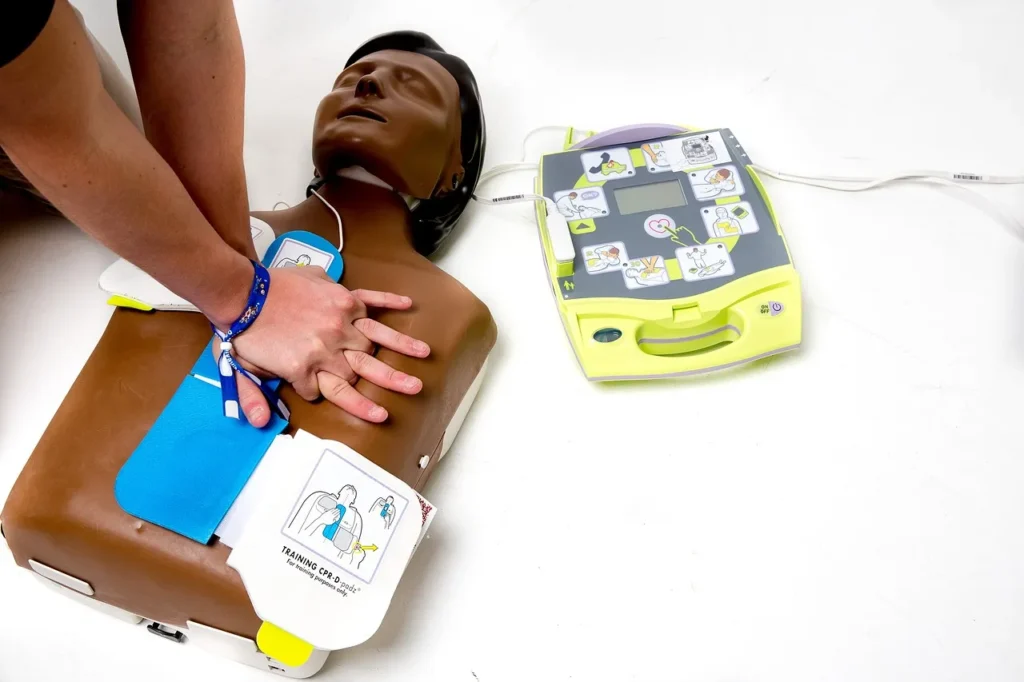
Lifepak devices are made for trained professionals like EMTs, paramedics, and healthcare providers. Some models, like the Lifepak CR Plus and CR2, are designed for public use. They feature automated guidance, making it possible for a layperson to help someone experiencing sudden cardiac arrest. Wearable or implantable devices are for high-risk patients, but Lifepak AEDs ensure help is available in public spaces and emergency vehicles.
How Lifepak Works
Lifepak monitors the heart’s rhythm and decides if a shock is needed. Automated models provide verbal instructions and prompts. To deliver a shock, pads or paddles are placed on the patient’s chest. Lifepak briefly stops the heart’s electrical activity so it can restart with a normal rhythm. Advanced models like the Lifepak 15 and 35 include CPR guidance, ensuring chest compressions continue while the device analyzes the heart.
After Using a Lifepak
After a shock, rescuers continue CPR while monitoring the patient. Lifepak devices record shocks, heart rhythms, and CPR timing. Some models, like the Lifepak 15 and 35, can transmit ECGs wirelessly to hospitals. If the first shock is not successful, additional shocks and medications may be administered under professional supervision. Lifepak helps bridge prehospital care to advanced hospital treatment.
Lifepak Benefits and Risks
Benefits: Lifepak can save lives if used promptly on the correct arrhythmias. Features like CPRInsight (in Lifepak 35 and CR2) support effective resuscitation, even in chaotic settings.
Risks: Using a defibrillator on the wrong arrhythmia can be harmful. Lifepak AEDs minimize this risk by automatically detecting shockable rhythms and providing step-by-step guidance.
Recovery and Follow-Up
After a cardiac event, patients may need weeks or months to recover. Lifepak devices support early intervention, increasing survival chances and improving outcomes. Survivors might require therapy or long-term monitoring. Regular check-ups ensure any implantable devices, like ICDs, function correctly alongside external Lifepak support in emergencies.
How Lifepak Saves Lives
The main goal of Lifepak defibrillators is simple: save lives during cardiac emergencies. When sudden cardiac arrest occurs, every second counts. Lifepak devices analyze a patient’s heart rhythm and deliver a shock when needed to restore normal rhythm. Many models also provide CPR guidance with metronome sounds and voice prompts, helping rescuers deliver effective compressions.
Advanced units go even further, offering continuous monitoring, oxygen saturation measurement, and synchronized cardioversion. By combining life-saving shocks with real-time feedback and monitoring, Lifepak gives both professionals and lay rescuers the tools they need to act quickly and confidently.
Latest Lifepak Technology
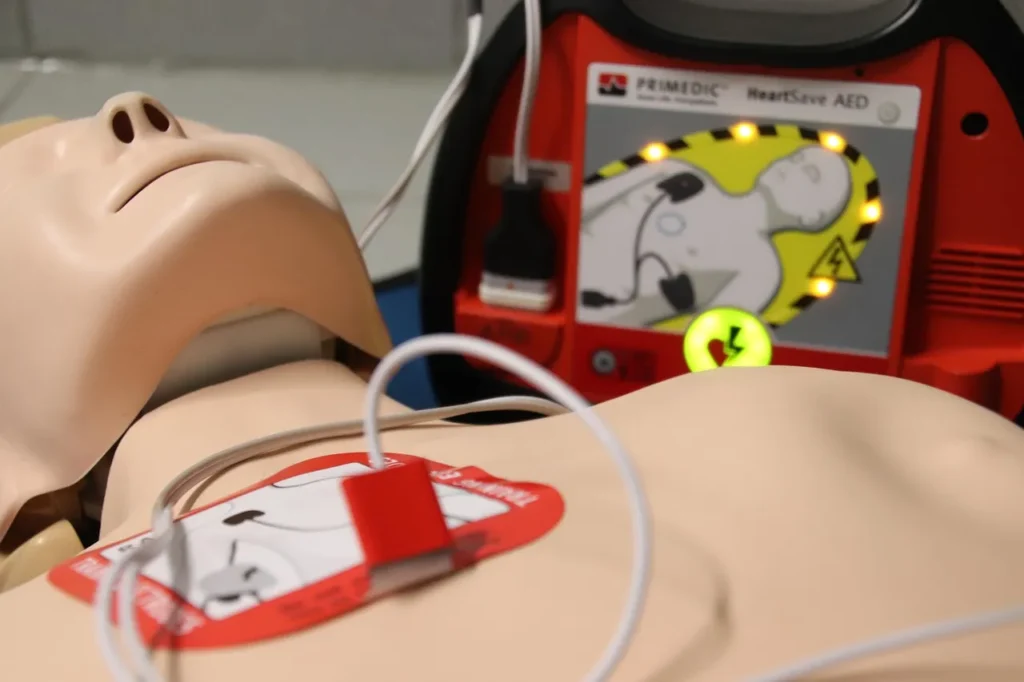
The newest generation of Lifepak devices showcases how far defibrillation technology has come. The Lifepak 35, introduced in 2024, is the most advanced model yet. It replaces many hard buttons with a touchscreen interface, making it easier and faster to use. It also includes CPRINSIGHT and STJInsight technologies, which allow rescuers to continue chest compressions while the device analyzes the heart rhythm—saving critical seconds during resuscitation.
In addition, the Lifepak 35 supports pediatric protocols, wireless data transfer, and diagnostic-quality ECG displays. This evolution highlights Lifepak’s commitment to innovation, ensuring that first responders and hospitals have access to cutting-edge tools that can truly make the difference between life and death.
Lifepak Models Comparison: From Early Units to Modern Devices
Over the years, Lifepak defibrillators have evolved from heavy, basic machines into lightweight, advanced, and user-friendly devices. Early models focused mainly on delivering shocks, while newer models combine defibrillation with monitoring, data transmission, and real-time CPR guidance. The table below highlights the key differences between some of the most notable Lifepak models.
| Model | Release Year | Key Features | Primary Use |
|---|---|---|---|
| Lifepak 33 | 1968 | First Lifepak model, 34 lbs, basic defibrillation | Hospitals & early EMS |
| Lifepak 5 | 1976 | Portable (under 6 lbs), used in Everest expeditions | Field EMS & remote use |
| Lifepak 12 | 1998 | Advanced monitoring, 12-lead ECG, reliable in ambulances | Emergency medical services |
| Lifepak 500 | 1997 | Public access AED, simple interface | Public use (trained responders) |
| Lifepak 15 | 2008 | Color LCD, CPR metronome, Bluetooth ECG transmission | Hospitals & ambulances |
| Lifepak 1000 | 2006 | Rugged AED, advanced protocols for professionals | Police, EMS, clinics |
| Lifepak 35 | 2024 | Touchscreen, CPRINSIGHT, pediatric AED modes, wireless data |
Who Should Use Each Lifepak Model?
Different Lifepak models are designed for specific users and environments. Hospitals and advanced EMS teams benefit from professional models like the Lifepak 15 and Lifepak 35, which offer full monitoring, 12-lead ECGs, and advanced features for critical care. First responders such as firefighters and paramedics often rely on the Lifepak 1000 for its rugged build and reliable AED functions in fast-moving emergencies.
For public spaces like schools, offices, and airports, the Lifepak 500 and Lifepak CR2 are ideal. These models are built for simplicity so trained non-medical users can act quickly in a cardiac emergency. This variety ensures that no matter the setting—from a hospital ICU to a shopping mall—there is a Lifepak device ready to save lives.
LIFEPAK in Popular Culture
LIFEPAK devices have even appeared in films. In the 1975 movie Three Days of the Condor, LIFEPAK 911 monitors are shown in an ICU scene.
Why LIFEPAK Matters
Cardiac arrest is a leading cause of death worldwide. Quick access to defibrillators can make the difference between life and death. LIFEPAK devices have made defibrillation more portable, more accurate, and more accessible.
With each new model, LIFEPAK improves the speed, safety, and effectiveness of emergency care. From mountaintops to city streets, these devices have helped save lives in the most critical moments.
Final Thoughts
LIFEPAK is more than just a line of defibrillators—it is a symbol of progress in medical technology. From the heavy LIFEPAK 33 to the modern LIFEPAK 35 with touchscreens and AI-driven analysis, the series continues to evolve.
Whether in the hands of paramedics, doctors, or trained civilians, LIFEPAK devices remain vital tools in the fight against sudden cardiac arrest.
Frequently Asked Questions (FAQ)
A Lifepak defibrillator is a medical device used to deliver an electric shock to a patient’s heart during cardiac arrest. Many models also monitor vital signs and provide guidance during CPR.
Lifepak devices are developed and produced by Physio-Control, now part of Stryker, a leading medical technology company.
No. Lifepak defibrillators are designed for different users. While advanced models are used by hospitals and paramedics, public access models like the Lifepak CR2 are simple enough for trained non-medical personnel.
Yes. Several models include pediatric AED protocols and accessories designed for safe use on children in cardiac emergencies.
The Lifepak 35 introduces a touchscreen interface, advanced diagnostic display, cprINSIGHT technology, and support for both 12-lead and 15-lead ECGs, making it one of the most advanced defibrillators available.
AEDs drastically improve survival rates from sudden cardiac arrest by providing immediate intervention before emergency services arrive. Every minute counts, and having a Lifepak nearby can save lives.